Pancreatic cancer is one of the most challenging cancers to diagnose and treat due to its aggressive nature and poor prognosis. A recent study published in Disease Models & Mechanisms titled “Targeted irradiation in an autochthonous mouse model of pancreatic cancer induces systemic changes in the tumor microenvironment and immune system” highlights a groundbreaking approach to imaging pancreatic tumors. The study demonstrates, for the first time, that a CT contrast agent, Fenestra HDVC, is taken up by pancreatic tumor cells, enabling direct tumor visualization via CT imaging.
Positive CT Contrast in Tumor
Traditionally, contrast-enhanced CT imaging of tumors has relied on vascular contrast agents, which highlight blood vessels rather than the tumor itself. However, this study reveals that Fenestra HDVC actively accumulates within pancreatic tumor cells instead of being restricted to vasculature. This suggests that tumor cells may be metabolizing, retaining, or passively accumulating the contrast agent, offering a new and more precise method for CT-based tumor detection.
Overcoming Challenges in Pancreatic Tumor Imaging
Pancreatic tumors are notoriously difficult to image with standard CT because they are often poorly vascularized and do not retain traditional contrast agents effectively. The ability of Fenestra HDVC to be retained in pancreatic tumors presents a new approach for early diagnosis and monitoring, potentially improving patient outcomes.
Potential Mechanisms of Uptake
The exact mechanism behind Fenestra HDVC uptake by pancreatic tumor cells remains unclear, but several hypotheses have been proposed:
- Metabolic alterations in cancer cells that increase contrast agent retention.
- Leakage of contrast agent into the tumor microenvironment, followed by uptake by tumor-associated macrophages or tumor cells.
- Changes in cell membrane permeability in pancreatic cancer cells allowing internalization of the contrast agent.
If this uptake pattern is reproducible, Fenestra HDVC could be developed into a novel tumor-targeting contrast agent for both preclinical and clinical pancreatic cancer imaging.
Evidence of Tumor-Specific Accumulation
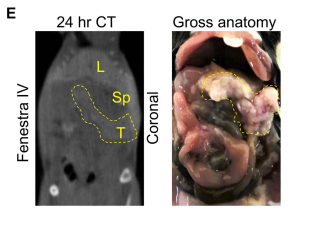
A key finding in the study is illustrated in Figure S1E, which shows the uptake of Fenestra HDVC in tumor tissue rather than blood vessels. At the specific time point shown, the contrast agent had already been cleared from circulation, demonstrating preferential accumulation in the tumor. This highlights the ability of Fenestra HDVC to specifically target tumors, providing a clearer and more accurate imaging contrast.
Implications for Cancer Diagnosis and Treatment
One of the features of this imaging approach is that Fenestra HDVC does not remain in the vasculature at later time points. This is in contrast to other contrast agents that may persist in blood vessels, making it difficult to distinguish between tumor tissue and blood supply in imaging. This distinction makes Fenestra HDVC a promising candidate for future cancer diagnostics.
Future Directions in Cancer Imaging
The success of Fenestra HDVC in imaging pancreatic tumors paves the way for more advanced applications of nanomedicine and biomarker-targeted imaging in cancer research. As further studies refine and test this method across other tumor models, there is great potential for its use in human clinical trials. If proven effective in clinical settings, Fenestra HDVC could revolutionize the early diagnosis and monitoring of pancreatic carcinoma, leading to improved survival rates and better prognostic tools.
Conclusion
This study represents a significant advancement in pancreatic cancer imaging, demonstrating how Fenestra HDVC could transform the way tumors are detected and monitored. By leveraging tumor-specific contrast accumulation, researchers and clinicians may soon have a powerful new tool in their fight against one of the most challenging cancers to treat.
Citation:
Tesson M, Stevenson K, Karim SA, Nixon C, Chalmers AJ, Sansom OJ, O’Neill E, Jones K, Morton JP. Targeted irradiation in an autochthonous mouse model of pancreatic cancer. Dis Model Mech. 2024 Mar 1;17(3):dmm050463. doi: 10.1242/dmm.050463. Epub 2024 Mar 14.
*This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format
*Fenestra LC named in the article is currently undergoing a name change to Fenestra HDVC.